How Do You Know the Streric Number of Atomic Orbital and Hybrid Chemistry
A Shortcut For Determining The Hybridization Of An Atom In A Molecule
Here's a shortcut for how to decide the hybridization of an atom in a molecule. This volition salve you a lot of time.
–Begin SHORTCUT–
Here's what you do:
- Look at the atom.
- Count the number of atoms connected to information technology (atoms – non bonds!)
- Count the number of alone pairs attached to it.
- Add together these two numbers together.
- If information technology'south 4, your cantlet is sp3.
- If it'south 3, your cantlet is sptwo.
- If it's 2, your cantlet is sp.
(If it's 1, information technology'southward probably hydrogen!)
This works in at least 95% of the cases y'all will see in Org ane.
–Stop SHORTCUT–
Tabular array of Contents
- Some Uncomplicated Worked Examples Of The Hybridization Shortcut
- How To Decide Hybridization Of An Atom: 2 Exercises
- Are There Any Exceptions?
- Exception #ane: Lone Pairs Adjacent To Pi-bonds
- Lone Pairs In P-Orbitals (Versus Hybrid Orbitals) Have Amend Orbital Overlap With Adjacent Pi Systems
- Exception #2. Geometric Constraints
- "Geometry Determines Hybridization, Non The Other Way Around"
- Notes
1. Some Simple Worked Examples Of The Hybridization Shortcut
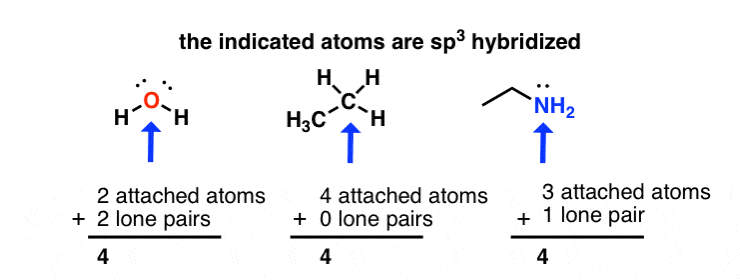
sp3 hybridization: sum of attached atoms + lone pairs = 4
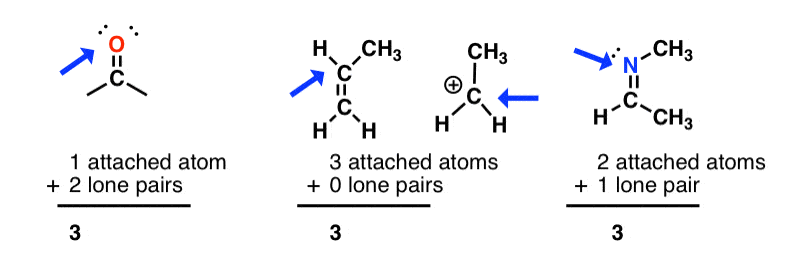
sp2 hybridization: sum of attached atoms + lone pairs = 3
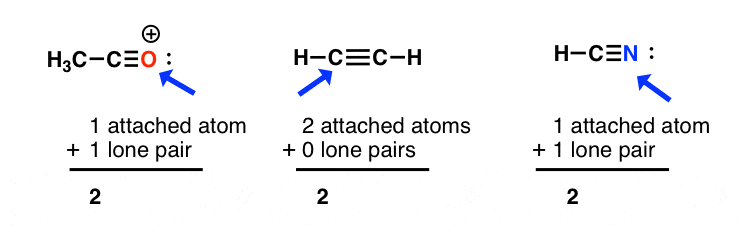
sp hybridization: sum of fastened atoms + lone pairs = 2
Where it tin can start to become slightly tricky is in dealing with line diagrams containing implicit ("hidden") hydrogens and lone pairs. Chemists like fourth dimension-saving shortcuts only as much as anybody else, and learning to quickly interpret line diagrams is equally fundamental to organic chemistry as learning the alphabet is to written English.
Remember:
- Just because alone pairs aren't drawn in on oxygen, nitrogen, and fluorine doesn't mean they're not there.
- Assume a full octet for C, Northward, O, and F with the post-obitane exception: a positive charge on carbon indicates that there are only six electrons around it. [Nitrogen and oxygen begetting a formal charge of +1 still have total octets].
[Advanced: a quick note on determining the hybridization of atoms in some weird cases like gratis radicals, carbenes and nitrenes ]
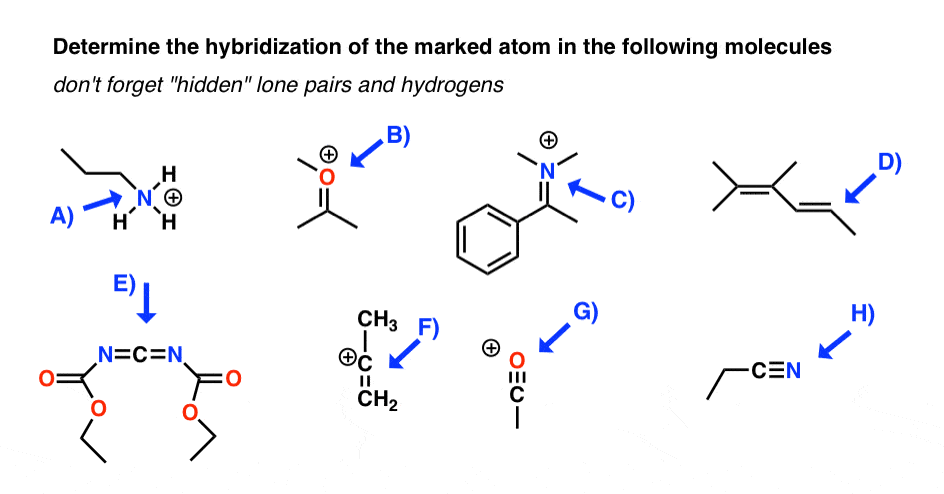
ii. How To Make up one's mind Hybridization Of An Cantlet: 2 Exercises
Here's an exercise. Try picking out the hybridization of the atoms in this highly poisonous molecule fabricated past the frog in funky looking pyjamas, beneath right.
[Don't worry if the molecule looks a little crazy: but focus on theindividual atoms that the arrows signal to (A, B, C, D, E). A and B especially. If y'all haven't mastered line diagrams nonetheless (and "subconscious" hydrogens) maybe get some more practise and come back to this later.]
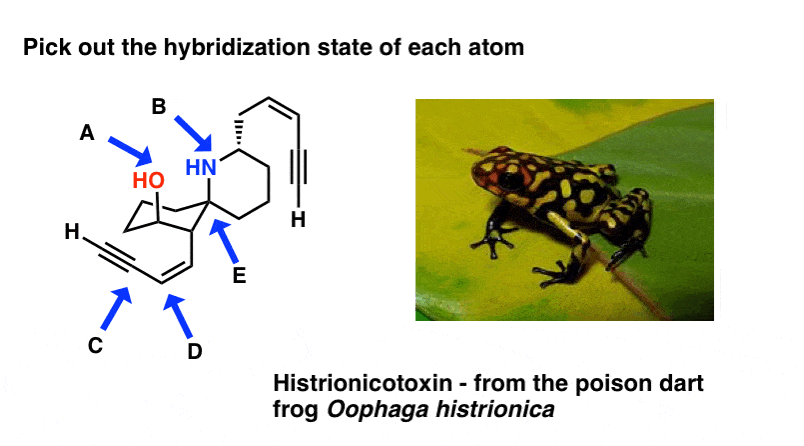
[Answers here]
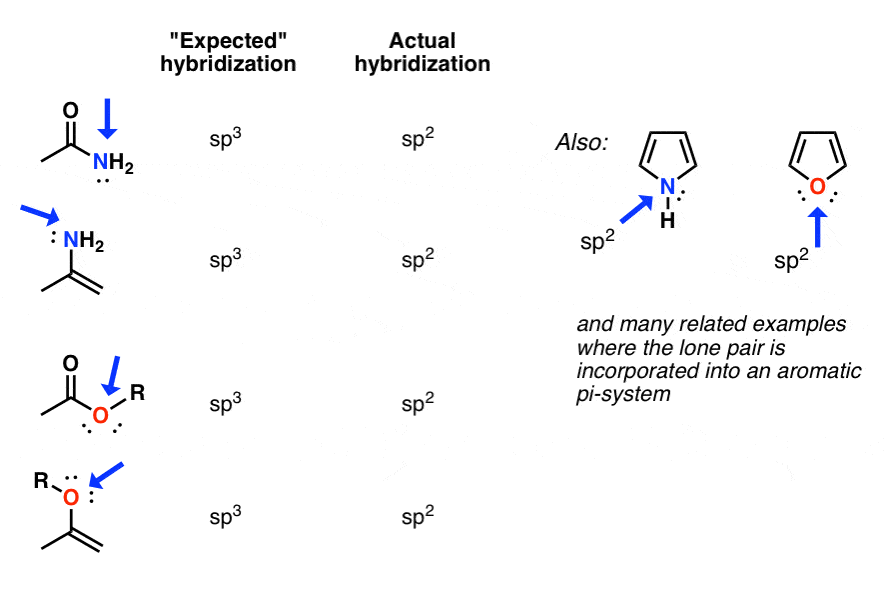
Here are some more than examples.
[Answers here.]
3. Are There Whatever Exceptions?
Sure. Although equally with many things, explaining the shortcut takes about two minutes, while explaining the exceptions takes about 10 times longer.
Helpfully, these exceptions fall into two main categories. Information technology should be noted that by the fourth dimension your class explains why these examples are exceptions, it will probable take moved far beyond hybridization.
Bottom line: these probably won't be establish on your start midterm.
4. Exception #1: Lone Pairs Next To Pi-bonds
The main exception is for atoms bearing lone pairs that are side by side to pi bonds.
Quick shortcut: Lone pairs side by side to pi-bonds (and pi-systems) tend to be in unhybridized p orbitals, rather than in hybridized spdue northorbitals. So when a nitrogen that you lot might expect to be trigonal pyramidal spiii is adjacent to a pi bond, its hybridization is actually sp2 (trigonal planar).
Why? The quick respond is that lowering of energy from conjugation more than than makes up for whatsoever gain in energy through increased steric hindrance. [encounter this postal service: "Conjugation and Resonance"]
What's the long answer?
5. Solitary Pairs In P-Orbitals (Versus Hybrid Orbitals) Have Better Orbital Overlap With Adjacent Pi Systems
Let's recall dorsum to why atoms hybridize in the first place: minimization of electron-pair repulsion.
For a primary amine like methylamine, adoption of a tetrahedral (sp3) geometry by nitrogen versus a trigonal planar (sp2) geometry is worth about 5 kcal/mol [roughly twenty kJ/mol]. That might not sound like a lot, but for two species in equilibrium, a departure of 5 kcal/mol in energy represents a ratio of about 4400:i] . [How do we know this? Meet this (advanced) Notation on nitrogen inversion]
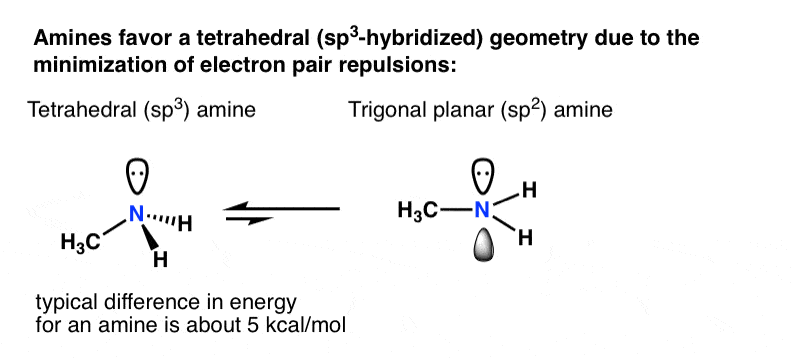
What if there was some compensating effect whereby a lonely pair unhybridized p-orbital was really more stable than if information technology was in a hybridized orbital?
This turns out to be the case in many situations where the lonely pair is side by side to a pi bond! The most common and of import example is that of amides, which institute the linkages betwixt amino acids. The nitrogen in amides is planar (sp2), not trigonal pyramidal (sp3), as proven by x-ray crystallography.
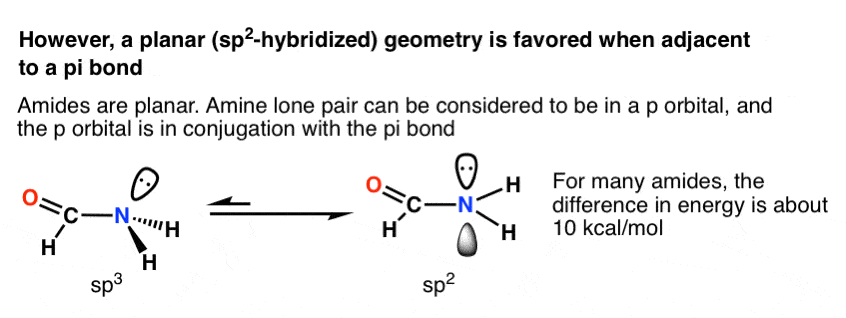
The difference in free energy varies widely, but a typical value is about 10 kcal/mol favouring the trigonal planar geometry. [Nosotros know this considering many amides have a measurable barrier to rotation a topic we too talked about in the Conjugation and Resonance post]
Why is trigonal planar geometry favoured here? Better orbital overlap of the p orbital with the pi bond vs. the (hybridized) sp3 orbital.
The cartoon beneath tries to bear witness how a change in hybridization from sp3 to sp2 brings the p-orbital closer to the adjoining p-orbitals of the pi bond, allowing for improve orbital overlap. Better orbital overlap allows for stronger pi-bonding between the nitrogen lone pair and the carbonyl p-orbital, which results in an overall lowering of energy.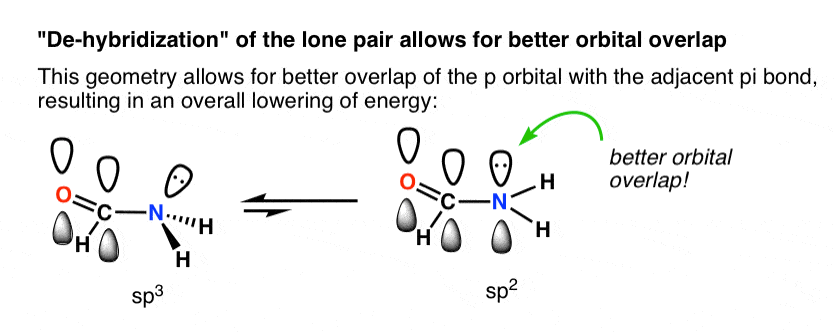
Y'all tin recollect of this as leading to a stronger "partial" C–N bond. 2 important consequences of this interaction are restricted rotation in amides, besides equally the fact that acid reacts with amides on the oxygen, not the nitrogen lone pair (!)
The oxygen in esters and enols is likewise also sptwo hybridized, every bit is the nitrogen in enamines and countless other examples.
Equally you will likely run across in Org 2, some of the most dramatic cases are those where the "de-hybridized" lone pair participates in an aromatic organization. Here, the energetic compensation for a change in hybridization from spiii to sp2 tin be very neat indeed – more than xx kcal/mol in some cases.
For this reason, the most basic site of pyrrole is not the nitrogen lone pair, but on the carbon (C-2) (!).
half-dozen. Exception #2. Geometric Constraints
Another instance where the actual hybridization differs from what we might look from the shortcut is in cases with geometric constraints. For instance in the phenyl cation below, the indicated carbon is attached two two atoms and zero lonely pairs. What's the hybridization?
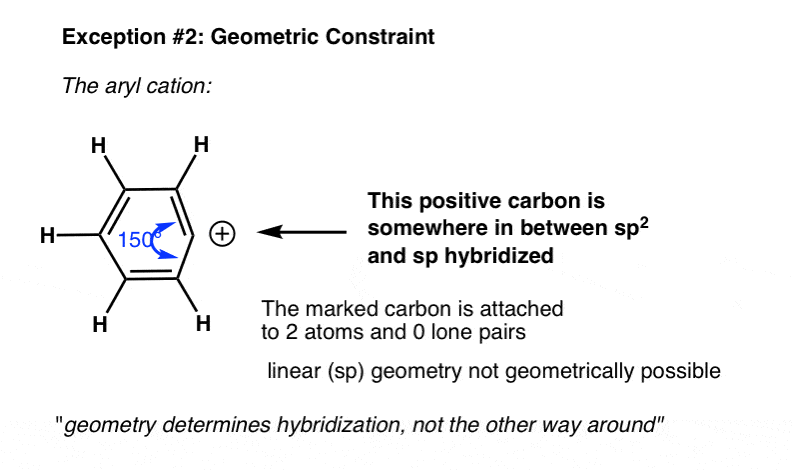
From our shortcut, we might expect the hybridization to be sp.
In fact, the geometry around the atom is much closer to sp2. That's considering the bending strain adopting the linear (sp) geometry would lead to far besides much angle strain to be a stable molecule.
7. "Geometry Determines Hybridization, Not The Other Way Around"
A quote passed on to me from Matt seems appropriate:
"Geometry determines hybridization, not the other way around"
Well, that'southward probably more than you wanted to know near how to make up one's mind the hybridization of atoms. Suffice to say, any post from this site that contains shortcut in the championship is a sure fire bet to have over 1000 words and >x figures.
Thank you to Matt Pierce of Organic Chemical science Solutions for of import contributions to this post. Ask Matt nearly scheduling an online tutoring session here.
Notes
Answers to Q1: A) sp3 B) spiii C) sp D) sp2 Eastward) spthree
Answers to Q2:A) spthree B) sptwo C) sp2 D) spii E) sp F) sp G) sp H) sp
Annotation 1. Some weird cases.
Unlikely to come across these, but here goes:
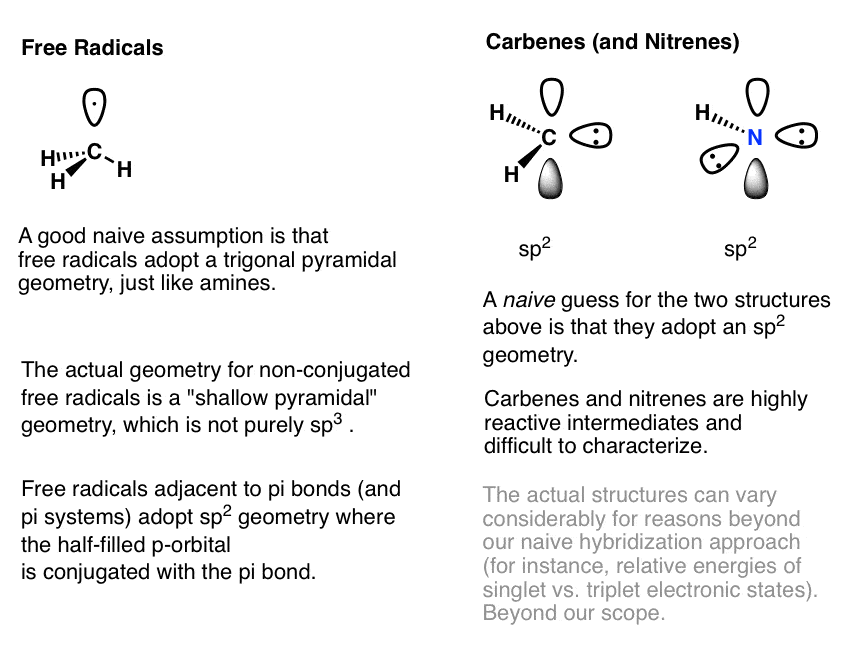
What virtually higher block elements similar sulfur and phosphorus?
Third row elements similar phosphorus and sulfur can exceed an octet of electrons past incorporating d-orbitals in the hybrid. This is more in the realm of inorganic chemistry so I don't really want to hash out information technology. Here's an instance for the hybridization of SF4 from elsewhere. (sp3d orbitals).
Note two: For the 5 kcal/mol effigy, come across here. [Tetrahedron Lett, 1971, 37, 3437].
An amine connected to three different substituents (R1 R2 and R3) should be chiral, since information technology has in full 4 different substituents (including the lone pair). Nonetheless, all early attempts to prepare enantiomerically pure amines met with failure. It was later plant that amines undergo inversion at room temperature, like an umbrella beingness forced within-out by a strong wind.
In the transition state for inversion the nitrogen is trigonal planar. One tin thus calculate the departure in energy betwixt the spiii and sp2 geometries by measuring the activation barrier for this process (notably by the work of Kurt Mislow (RIP)].
Note iii:A fun counter-instance might be Coelenterazine .
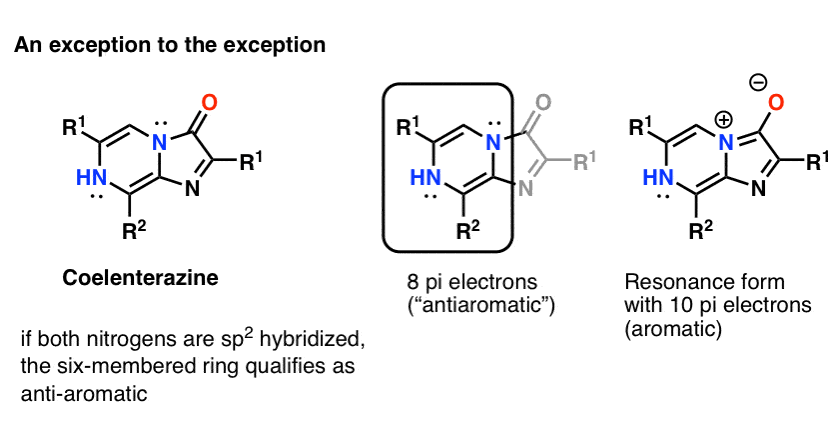
I would not expect both nitrogen atoms to be sp2 hybridized, considering that would lead to a cyclic, apartment, conjugated organization with viii pi electrons : in other words, antiaromatic. I tin can't notice a crystal structure of the core molecule to confirm (only would welcome any additional data!)
NOTE – (added afterward) If you lot draw the resonance form where the nitrogen lone pair forms a pi bond with the carbonyl carbon, so the ring system has 10 electrons and would therefore be "aromatic".
Source: https://www.masterorganicchemistry.com/2018/01/16/a-hybridization-shortcut/
0 Response to "How Do You Know the Streric Number of Atomic Orbital and Hybrid Chemistry"
Post a Comment